Atmospheric chemistry of reactive trace species through direct real-time measurements
Poor air quality represents the greatest environmental risk to public health in the UK (DEFRA, 2017), and has been reported to affect every organ in the body, with links to illnesses including cardiovascular and respiratory diseases, vascular dementia, and Alzheimer’s disease causing over 4.2 million premature deaths worldwide and costing the UK economy ~£15 billion each year. The properties and impacts of our air are governed by atmospheric composition, the understanding of which is vital to the development of policies aimed at improving air quality and requires knowledge of the emission rates, concentrations and chemistry of reactive trace species in the atmosphere.
Once released into the atmosphere, the dominant fate for the majority of hydrocarbons (HCs) and volatile organic compounds (VOCs) is oxidation by hydroxyl (OH) radicals, ozone (O3), or nitrate (NO3) radicals, leading to complex cascades of reactions which result in production of secondary pollutants including ozone and secondary organic aerosol (SOA), which are harmful to human health.
This project will investigate the chemistry and atmospheric impacts of reactive species using time-resolved ultraviolet and infrared absorption spectroscopy to make direct measurements of reactive atmospheric species in real-time during the course of their reactions. Laboratory experiments will focus on the chemistry of Criegee intermediates (CIs), a family of reactive species generated in the atmosphere through the reactions of ozone (O3) with unsaturated volatile organic compounds (VOCs) (Figure 1).
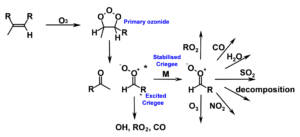
Figure 1: Production of Criegee intermediates in the atmosphere and their potential fates. Reaction kinetics and products of stabilised Criegee intermediate reactions are largely unknown and hinder our understanding of their impacts on atmospheric composition, air quality and climate.
Criegee intermediates react rapidly in the atmosphere, with potential impacts on the oxidising capacity of the atmosphere and formation of sulfate aerosol and secondary organic aerosol (Johnson and Marston, 2008; Khan et al., 2018). However, detailed assessments of the role and impacts of Criegee intermediates in the atmosphere are hindered by a lack of information regarding the production and subsequent chemistry of Criegee intermediates. A particular challenge since the proposal of the existence of Criegee intermediates in 1949 (Criegee & Wenner, 1949) has been their direct measurement owing to high reactivities and thus low concentrations. The advent of photolytic sources for Criegees, reported in 2012 (Welz et al., 2012), has facilitated laboratory studies of CIs and has enabled greater understanding of the chemistry and properties of Criegees, but there are still significant uncertainties regarding the kinetics and mechanisms of Criegee intermediate reactions under atmospherically relevant conditions.
Recent work in the Stone group has used photolytic methods to generate the Criegee intermediates CH2OO and CH3CHOO in the laboratory (Reactions R1-R4) coupled with time-resolved absorption spectroscopy to monitor Criegee intermediates in real-time during the course of a reaction (Figure 2) to determine their reaction kinetics (Mir et al., 2020; Onel et al., 2020; Onel et al., 2021; Mir et al., 2022).
CH2I2 + hν → CH2I + I (R1)
CH2I + O2 → CH2OO (R2)
CH3CHI2 + hν → CH3CHI + I (R3)
CH3CHI + O2 → CH3CHOO (R4)
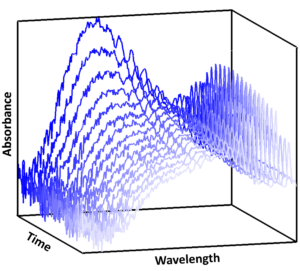
Figure 2: Time-resolved UV absorbance spectra used to monitor the Criegee intermediate CH2OO in real-time during its reaction with SO2. The Criegee intermediate is produced via photolysis of CH2I2/O2, with the maximum absorbance signal observed immediately following photolysis. The decay in absorbance is used to determine the reaction kinetics for CH2OO in the system (Mir et al., 2020).
Measurements of the kinetics of the Criegee intermediate CH2OO reactions with NO2 and SO2 within the group (Lade et al., manuscripts in preparation) have demonstrated discrepancies between predictions of the temperature dependence of these reactions and experimental measurements, highlighting the need for detailed investigation of further reactions to inform atmospheric models used to assess and predict air quality and climate.
This project will investigate the kinetics of key Criegee intermediate reactions in the atmosphere. Depending on your interests, you may also be involved in measurements of CIs and other key species in an atmospheric simulation chamber to explore the behaviour of Criegee intermediates directly, and quantitatively, in ozonolysis reactions using cavity enhanced absorption spectroscopy (CEAS).
Objectives
You will develop capabilities for direct real-time measurements of reactive species in the laboratory using time-resolved absorption spectroscopy during the course of reactions taking place on millisecond to microsecond timescales. This work will provide valuable information on the kinetics and mechanisms of Criegee intermediates for assessments of atmospheric composition.
You will have opportunities to develop skills in experimental design, kinetics, spectroscopy, atmospheric modelling and data analysis. You will develop experimental methodology that can be applied to a wide range of problems including those in atmospheric chemistry, combustion chemistry, astrochemistry, and materials chemistry.
This project links to the NERC objectives related to atmospheric pollution and human health, clean air, and climate.
Potential for high impact outcome
The role of chemistry in controlling atmospheric composition is of fundamental importance to our understanding of air quality and climate change. This work will provide valuable measurements of reaction kinetics and product yields to improve our understanding of atmospheric composition and chemistry, providing greater constraints on model calculations of global oxidising capacity and production of secondary organic aerosol. It is anticipated that this project will generate several papers, with potential for publication in high impact journals.
Training
The student will work under the supervision of Dr Daniel Stone and Professor Paul Seakins within the Atmospheric and Planetary Chemistry group in the School of Chemistry at the University of Leeds. You will be supported by a range of supervision from monthly meetings and group presentations, through to daily informal chats with supervisors. You will work in well-equipped laboratories and be part of an active, thriving and well-funded atmospheric chemistry community. The Leeds group has an internationally leading reputation in atmospheric chemistry for field measurements of atmospheric composition, laboratory studies of chemical kinetics and photochemistry, and the development of numerical models and chemical mechanisms. Activities in these three areas are intimately linked and interdependent, providing significant advantages. You will be supported to attend both national and international conferences, and will receive a wide range of training, for example in communication skills, project management, and with other technical aspects. The PhD will provide a wide range of experience in the use of high power lasers, vacuum systems, optics, computer controlled data acquisition systems and methods in numerical modelling. You will also have access to training provided by the National Centre for Atmospheric Science. The successful PhD student will have access to a broad spectrum of training workshops that include managing your degree and preparing for your viva (http://www.emeskillstraining.leeds.ac.uk/).
Student profile
The student should have an interest in atmospheric chemistry, air quality and global environmental problems, with a strong background in experimental physical chemistry or similar (e.g. physics, engineering, environmental science). Standard NERC eligibility rules apply.
References
Criegee & Wenner, Liebigs Ann. Chem., 564, 9-15, 1949
DEFRA, Department for Food and Rural Affairs, Air Pollution in the UK 2016, September 2017
Johnson and Marston, Chem. Soc. Rev., 37, 699-716, 2008
Khan et al., Environ. Sci. Processes Impacts, 20, 437-453, 2018
Mir et al., Phys. Chem. Chem. Phys., 17, 9448-9459, 2020
Mir et al., Atmos. Meas. Tech., 15, 2875-2887,2022
Onel et al., J. Phys. Chem. A, 124, 31, 6287-6293, 2020
