Nitrogen oxides in remote tropical ocean environments and their impact on air pollution and climate
Project summary
Nitrogen oxides (NOx ≡ NO+NO2) play a central role in the chemistry of the atmosphere, controlling both the production and loss of key air pollutants and climate gases such as methane, ozone, and particulate matter. Primarily emitted by human activity over land, NOx levels are typically very low in remote ocean environments, and yet still regulate key atmospheric processes in these environments. Due to the size of the Earth’s oceans, small changes in NOx levels can have large impacts on the atmospheres response to changing emissions. Recent work has highlighted significant apparent knowledge gaps in our understanding of NOx in remote ocean environments, which need to be addressed if we are to inform effective policies to tackle air pollution, climate and ecosystem health. This PhD project will use the custom built York NO-LIF instrument to make highly sensitive measurements of NOx in a remote tropical Atlantic environment, and use these data to advance our knowledge of this keystone of atmospheric chemistry.
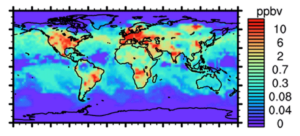 |
Figure 1: Surface NOx concentration calculated by the global atmospheric chemical transport model GEOS-Chem, showing low concentrations in remote ocean environments.
(Keller C. A. & Evans M. J. 2019) |
A major challenge in the study of NOx in remote ocean environments is that levels are so low that they are close to or below the limits of detection of current technologies (Fig.1). The Wolfson Atmospheric Chemistry Laboratories at the University of York have recently built a new instrument, based on laser-induced fluorescence (NO-LIF), for the highly sensitive and selective detection of NO. This instrument, the second of its kind in the world, enables measurements of NOx with previously unprecedented levels of accuracy and precision, and through comparison with data from state-of-the-science computation models will allow our understanding of this important chemistry to be challenged and improved.
This PhD project will involve the use of the York NO-LIF instrument as part of the NERC funded ConstrAining the Role of sulfur in the Earth System (CARES) project, which will make detailed atmospheric composition measurements in the North Atlantic region from both ship and aircraft platforms in 2025. The PhD project will be primarily based around the following objectives:
- Deploy highly sensitive instruments for the detection of Nitrogen oxides as part of the CARES field experiment aboard the UK FAAM 146 research aircraft and/or a NERC research vessel
- Use the data collected to investigate the sources and chemistry of NOx in the remote North Atlantic.
- Working with project collaborators, use the new data and understanding gained to challenge and improve our understanding of remote ocean atmospheric chemistry, through comparison with model predictions, and its impact on regional air pollution and climate.
This work will address an important knowledge gap in our understanding of atmospheric chemistry and thus directly improve our ability to design effective environmental policies.
Introduction
Nitrogen oxides (NOx ≡ NO + NO2) play a central role in atmospheric chemistry, regulating the chemical cycles that remove emitted pollutants, such as the climate gas methane, and produce secondary pollutants such as ozone and aerosols. A quantitative understanding of the sources, chemistry, and ultimate removal of NOx is therefore essential for the development of effective policies to tackle air pollution, climate and ecosystem health.
The importance of NOx means the study of processes that control its atmospheric abundance have been a major focus since the early days of atmospheric chemistry research. A recent study, however, showed that differences in the abundance and distribution of NO between current models used to predict air pollution and climate is the largest single uncertainty in predicted concentrations of the highly reactive hydroxyl radical, which in turn determines important factors such as the global methane lifetime (Zhou et al., 2019). Although differences between the models in this study were only small (of the order of single parts per trillion (ppt)), the high sensitivity of the chemistry to NOx indicates an important gap in our understanding of atmospheric chemistry.
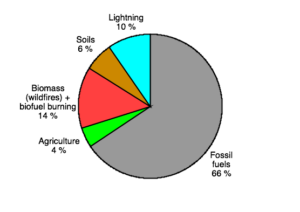 |
| Figure 2: Global NOx emissions. (IPCC AR4, Table 7.7) |
Global emissions of NOx are dominated by the anthropogenic burning of fossil fuels, with smaller natural sources including lightning and soil bacterial processes (Fig. 2). Once in the atmosphere, NOx has a lifetime of the order of days before it is converted into reservoir compounds. These NOx reservoirs are less reactive and thus have longer atmospheric lifetimes enabling them to be transported over large distances before they are either deposited, with impacts on ecosystem health, or broken down to re-release NOx. The continental dominance of NOx emissions means that in the remote marine atmosphere, a number of days transit time from the coast, we would expect NOx levels to be very low, and the inorganic nitrogen budget to be dominated by unreactive transported NOx reservoir species such as nitric acid. Although remote ocean NOx levels are significantly lower than over the continents, recent observations have found they are not as low as might be expected, indicating a potential missing NOx source (Ye et al, 2016). Figure 3 shows calculated (red) and measured (black) NO mixing ratios from a recent NASA aircraft mission over the remote Pacific. These differences between model and measured NO, of up to 5 ppt, have very significant impacts on the chemistry, and highlight that our understanding as represented in models needs to be improved.
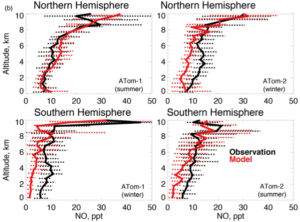 |
Figure 3: Plots of measured (black) and modelled NO mixing ratios over the remote Pacific Ocean during two recent NASA aircraft measurement campaigns. The large differences imply significant gaps in our understanding of this important chemistry in remote ocean regions.
(Travis et al. 2020) |
A key role for NOx in the atmosphere is as a catalyst for the oxidation and subsequent removal of other trace gases emitted from both natural and anthropogenic sources. One such process is the oxidation of the compound dimethyl sulphide (DMS), which is emitted by marine phytoplankton and oxidised to form sulfuric acid. This sulfuric acid is then able to create new atmospheric particles through a process called nucleation, and has important impacts on cloud formation in the remote marine atmosphere and is currently a major uncertainty in climate models. This process is the focus of the CARES project and the role of NOx in this chemistry will be the focus of this PhD.
A major challenge when studying remote ocean NOx chemistry is that the low abundance demands highly sensitive and selective measurements. Almost all of the research in the past two decades associated with direct detection of atmospheric NO has relied on the chemiluminescence detection technique. In this method, a sample of air is mixed with a high concentration of ozone resulting in the formation of electronically excited nitrogen dioxide which subsequently relaxes, releasing a detectable luminescence photon in the near-infrared. This technique while being reasonably precise (∼ 5-10 ppt detection limit), has several significant drawbacks. These include: potential interferences from other species; precision limits on the order of remote ocean NOx levels (5-10 ppt); significant instrumental background levels (10-100 ppt equivalent) which reduce accuracy; reliance on consumables including pure oxygen and dry ice; and production of %-levels of ozone which is toxic and must be exhausted from the instrument.
The recent development of a laser-induced fluorescence technique for the detection of NO (NO-LIF) has many advantages over CL (Rollins et al. 2020). The NO-LIF has detection limits of 0.3 ppt, has no known significant interferences, and is simple to operate. The Wolfson Atmospheric Chemistry Laboratories at the University of York has recently built an NO-LIF system, the second in the world. This project will use this instrument to address the current uncertainties surrounding remote ocean NOx, and thus provide crucial insight that will directly improve our ability to mitigate environmental risks such as air pollution and climate change.
The University of York (UoY) NO-LIF instrument is based on a detection system recently developed by collaborators at the US National Oceanic Atmospheric Administration (Rollins et al. 2020). The instrument itself is based around a custom-built fibre laser system, that capitalises on recent advances in telecommunications laser technology. This laser system enables the generation of 215 nm laser light which is used to excite NO molecules into an excited vibronic transition, from where they subsequently relax and we detect the resulting red shifted fluorescence from ∼ 255 – 267 nm (Fig. 4).
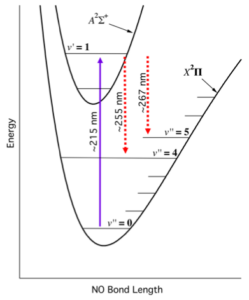
| Figure 4: Energy level schematic of NO-LIF system (Rollins et al. 2020). |
Project aims:
This PhD project will be focused on deploying the York NO-LIF instrument as part of the CARES project in the North Atlantic in 2025 and the subsequent analysis and interpretation of the data. There is the opportunity to deploy the instrument on both the FAAM 146 research aircraft and a NERC research vessel. Data from this deployment will then be analysed and interpreted using a range of techniques, including comparison with predictions from atmospheric chemistry models, to provide new insight into the processes controlling remote ocean atmospheric chemistry. The new science from this PhD project will directly impact the reliability of future air pollution and climate model predictions, and thus inform environmental policy.

| Figure 5: The FAAM 146 research aircraft. |
Skills
The instrumental and measurement focus of this PhD project means the successful candidate will have excellent practical / physical laboratory skills as well as a background in the physical sciences (e.g. Chemistry, Physics). The candidate will be enthusiastic about hands-on lab work, and show interest in environmental and/or public health issues. Computer programming experience, for both instrument control and data analysis, will be advantageous but significant training will be provided as part of the project (see Training section below). The Wolfson Atmospheric Chemistry Laboratories are a world class research centre, with significant technical support and resource for instrument development and data analysis. We appreciate that this PhD project encompasses several different science and technology areas, and we don’t expect applicants to have experience in many of these fields. The project is very well supported with experienced scientists and training in the new techniques and disciplines is all part of the PhD.
Training
The student will work under the supervision of Dr Pete Edwards and Prof James Lee and will be based at the Department of Chemistry’s Wolfson Atmospheric Chemistry Laboratory at the University of York. The Wolfson Atmospheric Chemistry Laboratories are home to more than 65 researchers with interests in all aspects of atmospheric chemistry, from stratospheric ozone, through to urban pollution, personal exposure and health. The laboratories comprise more than 1200 m3 of new offices and labs, the most recent extension to the building being completed in April 2018. The labs support an exceptional environment for research, have access to state-of-the-art facilities and include a range of different disciplines and researchers. The student will receive training on the NO-LIF instrument as well as techniques used to calibrate and characterise the instrument. Training will also be given on the computational skills needed to collect and analyse the data from the instrument. The University of York and the wider NERC PANORAMA DTP provide comprehensive training programmes for PhD students with a range of courses on both hard (e.g. data carpentry) and soft (e.g. presentation) skills. The student will also have access to training provided by the UK National Centre for Atmospheric Science such as the Introduction to Atmospheric Science course and Atmospheric Measurement Summer School on the Isle of Arran, and the Scientific Computing Course. The student will have the opportunity to present their work to the scientific community at national and international meetings and conferences, and will also be encouraged to take part in outreach events in order to disseminate the research beyond the immediate scientific community (e.g. to policymakers and the general public).
References
Keller, C. A. and Evans, M. J.: Application of random forest regression to the calculation of gas-phase chemistry within the GEOS-Chem chemistry model v10, Geosci. Model Dev., 12, 1209–1225, https://doi.org/10.5194/gmd-12-1209-2019, 2019.
Zhao, Y., et al.: Inter-model comparison of global hydroxyl radical (OH) distributions and their impact on atmospheric methane over the 2000–2016 period, Atmos. Chem. Phys., 19, 13701–13723, https://doi.org/10.5194/acp-19-13701-2019, 2019.
Denman, K.L., et al.: Couplings Between Changes in the Climate System and Biogeochemistry. In: Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA. 2007.
Ye, C., Zhou, X., Pu, D. et al. Rapid cycling of reactive nitrogen in the marine boundary layer. Nature 532, 489–491, https://doi.org/10.1038/nature17195, 2016.
Travis, K. R., et al.: Constraining remote oxidation capacity with ATom observations, Atmos. Chem. Phys., 20, 7753–7781, https://doi.org/10.5194/acp-20-7753-2020, 2020.
Wang X. T., et al.: Natural forcing of the North Atlantic nitrogen cycle in the Anthropocene, Proc. Natl. Acad. Sci. 115 (42) 10606-10611, https://doi.org/10.1073/pnas.1801049115, 2018.
Rollins, A. W., et al.: Single-photon laser-induced fluorescence detection of nitric oxide at sub-parts-per-trillion mixing ratios, Atmos. Meas. Tech., 13, 2425–2439, https://doi.org/10.5194/amt-13-2425-2020, 2020.
